
The Next Leap in Translational Neuroscience
At Transpharmation, our preclinical platforms continue to advance in step with the evolving challenges of CNS drug discovery and development. This month, we’re spotlighting recent progress across the following areas:
- Opioid-Induced Hyperalgesia and Opioid-Induced Tolerance
- Psychedelic Treatment to Improve Cognition
- Psychedelic Treatment for Alcohol Use Disorder
If you’re looking to strengthen the translational foundation of your next CNS program, these may provide meaningful scientific insight.

Opioids are supposed to relieve pain. Sometimes, they make it worse.
Opioid-Induced Hyperalgesia (OIH) and Opioid-Induced Tolerance (OIT) are two of the most clinically underestimated consequences of long-term opioid therapy, and two of the most challenging to address in drug development.
OIH is paradoxical by nature: the patient becomes more pain-sensitive under sustained opioid exposure. OIT progressively erodes analgesic efficacy, fueling dose escalation and its cascade of risks. Both phenomena share overlapping mechanisms (NMDA receptor upregulation, μ-opioid receptor desensitization, spinal sensitization, neuroinflammatory dysregulation) and both contribute directly to treatment failure and opioid misuse.
At Transpharmation, we have developed and validated two distinct mouse models to support drug discovery in this space:
- OIH model: BID morphine induction (10 mg/kg, D0–D4), compound testing on D6–D7. Dual endpoints: mechanical allodynia (von Frey) + thermal allodynia (hot plate). Positive controls: Methadone & Buprenorphine.
- OIT model: BID morphine induction (10 mg/kg, D0–D2), acute morphine challenge on D3–D4 to track attenuation of peak analgesic response. Positive controls: Ketamine & Dexmedetomidine.
Both models deliver reproducible pharmacological windows, suitable for MoA differentiation, compound screening, and dose-response characterization.
View Pain Models & Assays
Can Low Doses of Psychedelics Improve Motivation and Cognition?
Our published work explores this question using sub-threshold doses that do not induce psychedelic-like behavioral effects in rodents. Using large cohorts of male Long Evans rats trained in operant behavioral assays and segregated by performance level, we evaluated the effects of low doses of ketamine and psilocybin on key depression-related endophenotypes:
- Anhedonia & motivation: assessed using the Progressive Ratio (PR) task
- Attention & inhibitory control: assessed using the 5-Choice Serial Reaction Time Task (5-CSRTT)
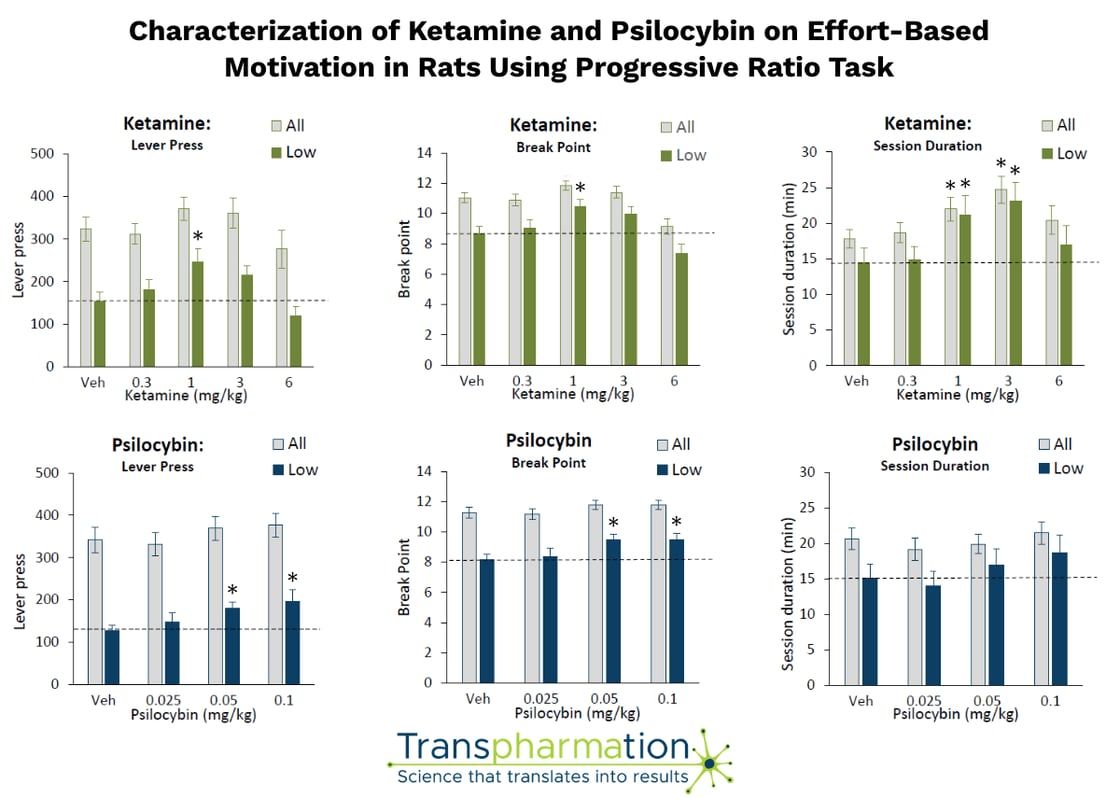
Key findings:
- ✔Low doses of ketamine (1–3 mg/kg) and psilocybin (0.05–0.1 mg/kg) improved motivation measured by increased breakpoints in the PR task
- ✔Equivalent doses of ketamine and psilocybin enhanced attentional accuracy and reduced impulsive action in the 5-CSRTT
- ✔Effects were modest but consistent, particularly in low-performing animals, suggesting a potential relevance for deficit-based models of psychiatric disease
- ✔These findings provide an important preclinical framework for studying low-dose psychedelic exposure, supporting the idea that sub-perceptual doses may still produce measurable functional benefits
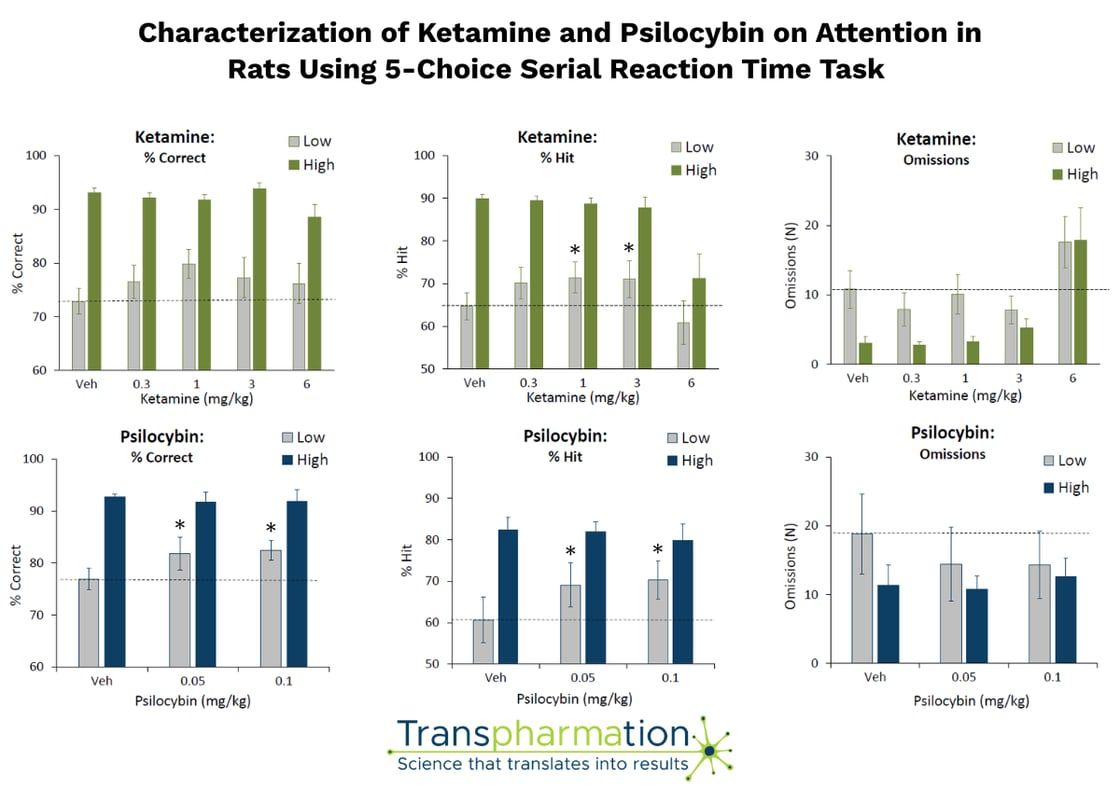
At Transpharmation, we leverage validated operant platforms to investigate domains such as motivation, attention, impulsivity, and cognitive flexibility — critical endpoints for psychiatric drug development.
Explore Operant Platform
Can Psychedelics Treat Alcohol Use Disorder?
Whether you are developing novel therapeutics for alcohol use disorder (AUD) or have a compound that may hold a secondary indication for AUD (e.g., GLP-1 agonists, psychedelics), our preclinical platform can help you generate the actionable, translational data you need.
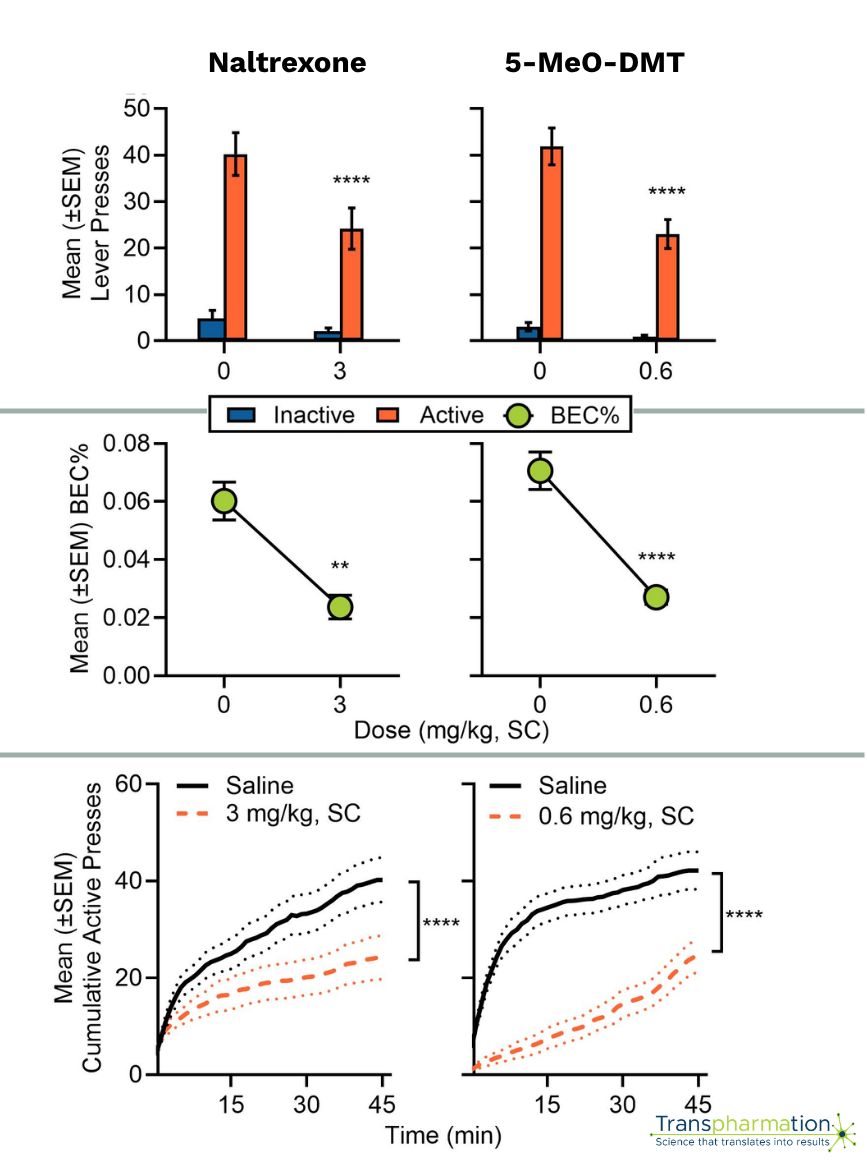
Transpharmation offers operant oral ethanol self-administration models that measure voluntary ethanol intake and associated blood ethanol concentration (BEC%) to evaluate the therapeutic potential of novel compounds for AUD.
Our model is sensitive to both naltrexone, a first-line pharmacological therapy for AUD, and the potent, fast-acting psychedelic 5-MeO-DMT, which both reduced ethanol intake and blood ethanol concentration in both sexes when administered prior to self-administration sessions.
Meet with us at these upcoming conferences:
- Hanson & Wade GLP-1-Based Therapeutics Summit
- Boston, MA, USA
- April 14–16, 2026
- OARSI — Osteoarthritis Research Society International
- West Palm Beach, FL, USA
- April 23–26, 2026
- CEBioForum
- Warsaw, Poland
- April 22–23, 2026
- Ewa Sokołowska is a panelist for "Smarter Science: Quo Vadis Testing"
- Hanson & Wade Neuroscience Innovation Partnering & Licensing Summit
- Boston, MA, USA
- April 28–29, 2026
- Pain Therapeutics Summit
- Amsterdam, Netherlands
- May 7–8, 2026
- Pain Mechanisms and Therapeutics Conference
- Verona, Italy
- May 17–22, 2026
- CPDD — College on Problems of Drug Dependence General Meeting
- Portland, OR, USA
- June 13–17, 2026
- Briana Renda will present "5-MeO-DMT and Naltrexone Selectively Attenuate Oral Ethanol Self-administration in Male and Female Wistar Rats"
- Briana Renda and Guy Higgins will also present a collaborative workshop with Altasciences: "Driving without a Roadmap: Addressing Regulatory Gaps and Translational Opportunities in Assessing CNS Drug Impairment"
- RSA — Research Society on Alcohol Annual Meeting
- San Antonio, TX, USA
- June 20–24, 2026
- the best validated assays to identify drugs for the quickest time to market
- bespoke laboratories with custom capabilities to suit your needs